Electronic Records – CSV FDA Warning Letters

Observation We remain concerned that your (b)(4) adverse drug experience reporting system has not been fully validated, and may have resulted in inaccurate assessment and untimely submission of 15-day alerts. The current application was released into production on November 9, 2009 using an Interim Validation Report (IVR) that is still not final. Critical issues (deviations) […]
FDA Observation of Excel Spreadsheets Validation
Observation 1. Your firm has not established and documented the accuracy, reliability and performance of your computer systems employed in the release of drug products [21 C.F.R. 211.68 (a)] For example, your firm did not verify the accuracy of Excel spreadsheets used to calculate product assay analytical results, for all products manufactured for […]
FDA recommendation on Deleting Electronic Data

Hybrid Systems (Deleting Electronic Data) In Hybrid systems the computerized system is used to generate a record which is printed and hand signatures applied. The decision for the regulated company is whether the electronic record (or raw data) should be kept or deleted from the system. In the FDA Guidance “Part 11, Electronic Records; […]
Computer Software Assurance (CSA) : Critical Thinking and Risk Based Approach

The Purpose of this Blog is to Provide guidance for the industry on the key concepts of Computer Software Assurance and provide direction to apply rational and critical thinking and commensurate it with risk associated with product quality and patient safety. This document takes into consideration challenges with the current computerized system validation approach and […]
Pharmacovigilance
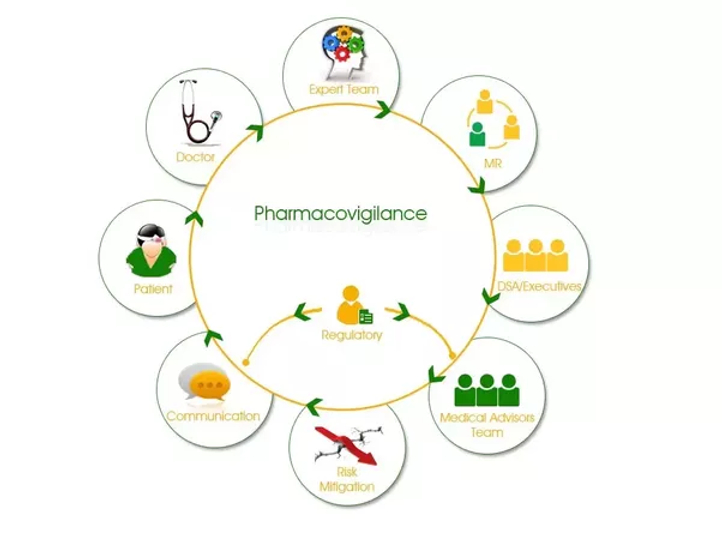
Knowing What Pharmacovigilance is? According to WHO Pharmacovigilance is the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other medicine/vaccine related problem. But to make it simple, etymologically Pharmacovigilance is to keep a watch on drugs (Pharmakon= Drug, Vigilare= to keep a watch). […]
Clinical Drug Development process and it’s Online Training

Developing a new drug from original idea to the launch of a finished product is a complex process which can take 12–15 years and cost in excess of $1 billion. A drug discovery programme initiates because there is a disease or clinical condition without suitable medical products available and it is this unmet clinical need […]
Computer System Validation (CSV) In Pharmaceutical Industry

Computerized system validation (CSV) (usually referred to as “Computer Systems Validation”) is the process of testing/validating/qualifying a regulated (E.g., FDA 21CFR11) computerized system to ensure that it does exactly what it is designed to do in a consistent and reproducible manner that is as safe, secure and reliable as paper records. This is widely used […]
Pharmacovigilance: Post Pandemic

Get Best training and certification in Pharmacovigilance The History of Pharmacovigilance roots back to the rise in awareness among clinicians and the public pertaining to the safety of drug use in the middle of 20th century. Despite concerns around health issues due to adverse reactions caused by intake of medicines, the discipline and practice […]
Medical Writing
Become a certified Medical writer from best Institute in India Changing market dynamics, expiring patents, and depleted product pipelines are all exerting pressure on pharmaceutical companies to outsource non-core activities in an effort to control costs and enhance drug-development cycle time. One of these activities is medical writing—an area that, although not at the core […]
Herbal Drug Regulatory Affairs

Herbal medicines, known as phytomedicines or botanical medicines (Phyto- Plant) contains plant parts like, leaves, roots, stem, flowers, and seeds for therapeutic purpose. It is the oldest and still most generally used system of medicine in the world at present. 80% of the world population relies on herbal medicines as their primary healthcare system. Get […]
