In the pharmaceutical industry, compliance is not just a requirement—it’s part of everyday operations. Every system, every record, and every process must be accurate, secure, and ready for inspection. This is where Computer System Validation (CSV), data integrity, and regulatory compliance play a crucial role.
Managing these activities separately often leads to confusion, delays, and compliance gaps. That’s why many organizations now prefer a one-stop solution—a single partner who can handle everything efficiently from start to finish.
SkillBee Solutions supports this approach by providing integrated CSV and compliance services tailored to the needs of pharma companies.
Why a One-Stop Approach Works Better

When validation and compliance activities are managed in one place, the process becomes more structured and efficient. There is better coordination, consistent documentation, and fewer chances of errors.
With the support of SkillBee Solutions, organizations can focus on their core operations while ensuring all validation activities are aligned with regulatory expectations
What Does a Complete CSV Service Include?
A complete CSV solution covers the entire system lifecycle in a practical and structured way.
1. Documentation and Planning
Every validated system starts with clear documentation such as:
- User Requirement Specification (URS)
- Functional and Design Specifications
These documents define how the system should work and create a strong foundation for validation. With SkillBee Solutions, documentation is always structured and audit-ready.
2. Risk-Based Validation
Modern validation follows a risk-based approach, focusing on critical areas instead of everything equally.
This includes:
- Identifying high-risk system functions
- Performing assessments like HLRA
- Prioritizing critical processes
SkillBee Solutions ensures that validation efforts are both efficient and compliant.
3. Testing and Qualification (IQ, OQ, PQ)
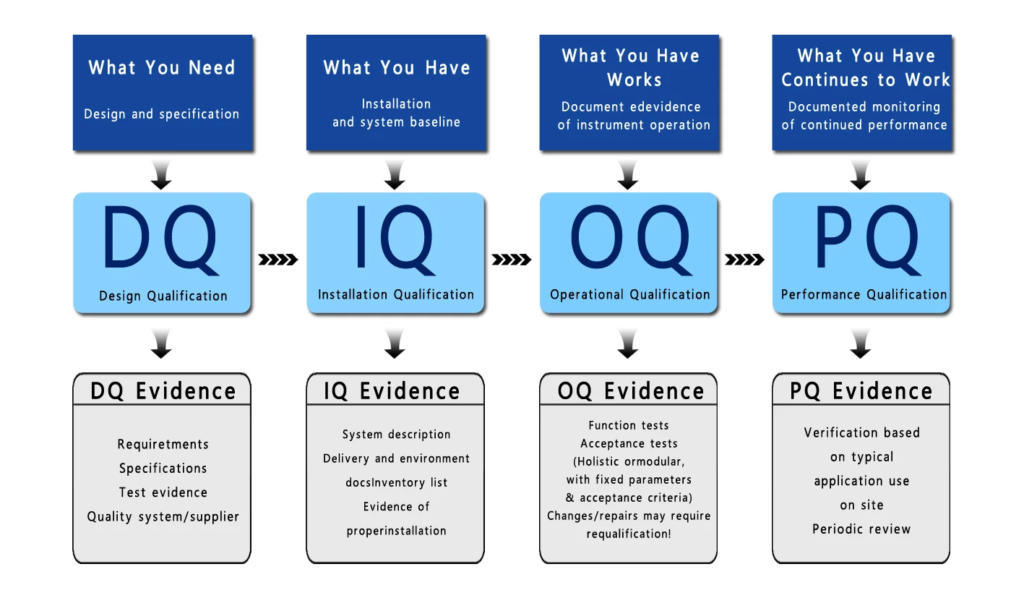
Systems are verified through:
- IQ: Installation check
- OQ: Functional verification
- PQ: Performance confirmation
These steps ensure that systems work correctly in real conditions, with proper support from SkillBee Solutions.
4. Data Integrity and Compliance
Data must remain accurate, consistent, and secure throughout its lifecycle.
Key focus areas include:
- Audit trail review
- Implementation of ALCOA+ principles
- Controlled data management
SkillBee Solutions helps maintain strong data integrity practices aligned with regulatory standards.
5. IT Infrastructure and System Support
A stable IT environment is essential for validated systems.
Services include:
- Infrastructure qualification
- Server and network validation
- Support for Cloud-based system
With SkillBee Solutions, organizations get a reliable and compliant IT foundation.
6. Audit Readiness
Being prepared for audits is critical in pharma.
A structured approach ensures:
- Proper documentation
- Quick audit responses
- Confidence during inspections
SkillBee Solutions helps companies stay audit-ready at all times.
Benefits of Choosing the Right Partner
Working with a single CSV service provider brings clear advantages:
- Better coordination across all activities
- Faster execution and reduced delays
- Improved compliance with global standards
- Less stress during audits
By partnering with SkillBee Solutions, organizations gain a dependable support system for all validation and compliance needs.
Conclusion
Managing Computer System Validation, data integrity, and regulatory compliance doesn’t have to be complex. With a structured approach and the right partner, it becomes a smooth and efficient process.
A one-stop solution—like the one offered by SkillBee Solutions—brings everything together, helping organizations stay compliant, organized, and future-ready.






