DRUG REGULATORY AFFAIRS
Drug Regulatory Affairs refers to all aspects within the pharmaceutical development process and how they are subject to various degrees of regulation. The pharmaceutical law frame, guidelines covering Quality, Safety and Efficacy as well as Health Authorities’ attitudes and requirements etc. have a great influence on the drug development process and the success of it. […]
Computerized System Validation

Computer System Computer hardware components assembled to perform in conjunction with a set of software programs, which are collectively designed to perform a specific function or group of functions. (PIC/S, PI 011-3, 25 September 2007) Computerized system:- A computer system consists of hardware, software, and network components, together with the controlled functions and associated […]
Basics of Project Management

Hello Readers, In today’s fast-paced developing professional world, every individual should be at par with the most required soft skill i.e. management. Management comes with a huge umbrella ranging from time management, resource management, skill management, etc. All these aspects converge to cover what is commonly called project management. Project management is […]
Change Management

Hello Readers, Hope it was a great weekend. And now as the night grows darker, and the day prepares to change from Sunday to Monday, there’s this little anxiety of the change. Talking of the change, let us now dig a deeper in the change management requirements during project management or in any industry […]
Industry 4.0 & Pharmaceutical Manufacturing : Digital Factory of Next Generation

The term Industry 4.0 refers to the fourth industrial revolution which brings together rapidly evolving technologies such as the internet of things(IoT),artificial intelligence(AI),robotics, and advanced computing to dramatically change the landscape of manufacturing. Industry 1.0 Industry 1.0 saw the manual processing of botanical, mineral, and animal derived materials transition from simple hand-operated tools to […]
Electronic Records – CSV FDA Warning Letters

Observation We remain concerned that your (b)(4) adverse drug experience reporting system has not been fully validated, and may have resulted in inaccurate assessment and untimely submission of 15-day alerts. The current application was released into production on November 9, 2009 using an Interim Validation Report (IVR) that is still not final. Critical issues (deviations) […]
FDA Observation of Excel Spreadsheets Validation
Observation 1. Your firm has not established and documented the accuracy, reliability and performance of your computer systems employed in the release of drug products [21 C.F.R. 211.68 (a)] For example, your firm did not verify the accuracy of Excel spreadsheets used to calculate product assay analytical results, for all products manufactured for […]
FDA recommendation on Deleting Electronic Data

Hybrid Systems (Deleting Electronic Data) In Hybrid systems the computerized system is used to generate a record which is printed and hand signatures applied. The decision for the regulated company is whether the electronic record (or raw data) should be kept or deleted from the system. In the FDA Guidance “Part 11, Electronic Records; […]
Computer Software Assurance (CSA) : Critical Thinking and Risk Based Approach

The Purpose of this Blog is to Provide guidance for the industry on the key concepts of Computer Software Assurance and provide direction to apply rational and critical thinking and commensurate it with risk associated with product quality and patient safety. This document takes into consideration challenges with the current computerized system validation approach and […]
Pharmacovigilance
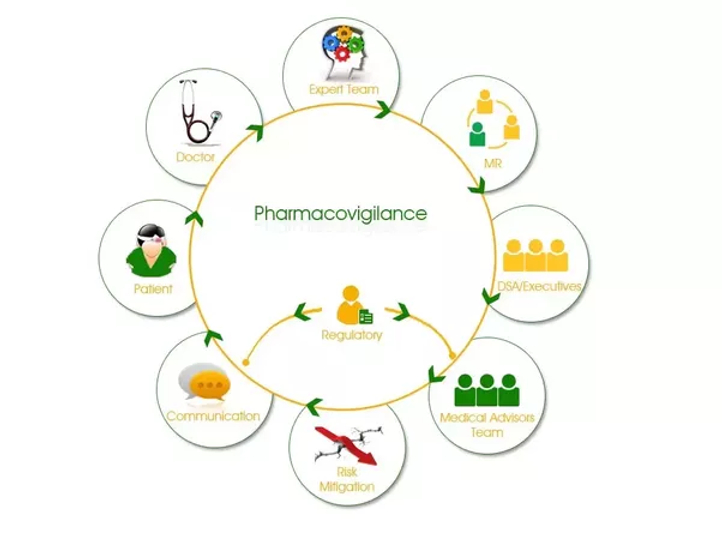
Knowing What Pharmacovigilance is? According to WHO Pharmacovigilance is the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other medicine/vaccine related problem. But to make it simple, etymologically Pharmacovigilance is to keep a watch on drugs (Pharmakon= Drug, Vigilare= to keep a watch). […]
