The journey of a drug from laboratory discovery to reaching patients is long, complex, and highly regulated. The drug approval process ensures that every medicine available in the market is safe, effective, and of high quality.
In the pharmaceutical industry, drug regulatory affairs plays a crucial role in managing approvals and compliance with authorities like the Food and Drug Administration and Central Drugs Standard Control Organization.
If you are a student or professional, understanding this process is essential for building a career in pharma and clinical research.
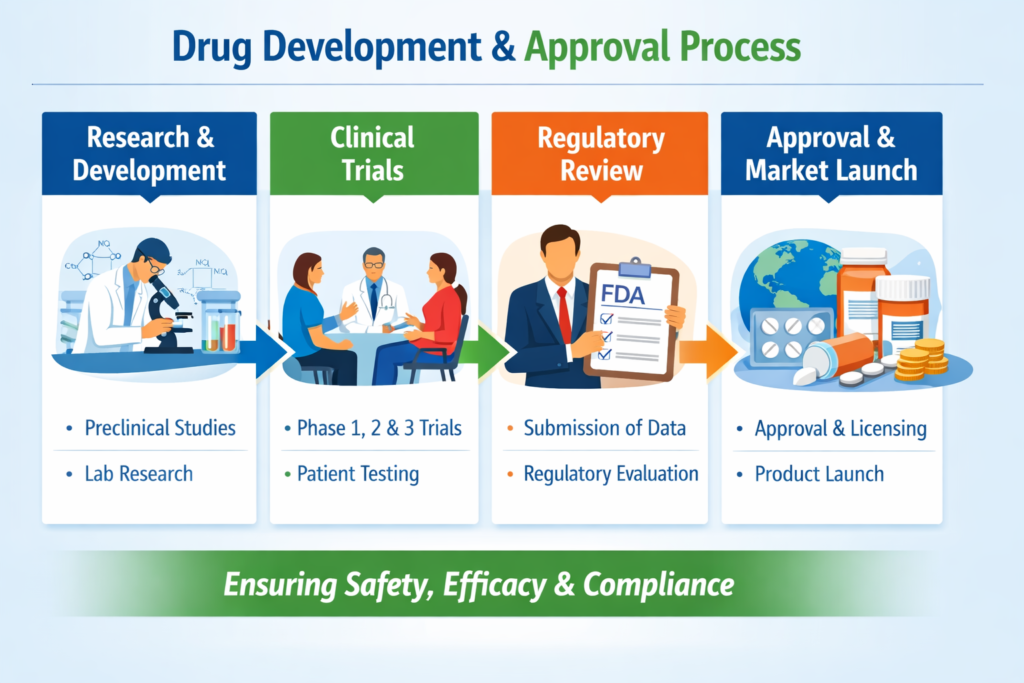
Step 1: Drug Discovery and Preclinical Research
The process begins in the laboratory, where scientists identify a potential drug candidate. This stage includes:
- Target identification
- Laboratory experiments
- Animal testing (preclinical studies)
The main goal is to evaluate safety and biological activity before testing on humans.
Step 2: Clinical Trials (Human Testing)
Once preclinical studies are successful, the drug enters clinical trials, which are conducted in three phases:
Phase 1
- Conducted on a small group of healthy volunteers
- Focus: Safety and dosage
Phase 2
- Conducted on patients
- Focus: Effectiveness and side effects
Phase 3
- Large-scale trials
- Confirms safety and efficacy
Clinical trials are strictly regulated to ensure patient safety and data accuracy.
Step 3: Regulatory Submission (NDA/BLA Filing)
After successful clinical trials, the pharmaceutical company submits all data to regulatory authorities.
This submission is called:
- NDA (New Drug Application)
- BLA (Biologics License Application)
It includes:
- Clinical trial data
- Manufacturing details
- Safety reports
Regulatory teams prepare CTD/eCTD dossiers, which are standardized formats used globally.
Authorities like the European Medicines Agency also review such submissions for approval in Europe.
Step 4: Regulatory Review and Approval
Once submitted, regulatory bodies carefully review the application.
Authorities evaluate:
- Drug safety
- Effectiveness
- Manufacturing quality
If everything meets standards, the drug receives marketing approval.
If not, companies may be asked for additional data or clarification.
Step 5: Manufacturing and Quality Control
After approval, the drug enters mass production.
Key activities include:
- Large-scale manufacturing
- Quality Assurance (QA)
- Quality Control (QC)
Companies must follow Good Manufacturing Practices (GMP) to ensure consistency and safety.

Step 6: Market Launch and Post-Marketing Surveillance
Finally, the drug is launched in the market.
But the process doesn’t end here. Continuous monitoring is done through:
- Pharmacovigilance
- Adverse drug reaction reporting
- Post-marketing studies
This ensures long-term safety of the drug.
Role of Regulatory Affairs in Drug Approval
Regulatory affairs professionals act as a bridge between pharma companies and regulatory authorities.
Their responsibilities include:
- Preparing documentation
- Ensuring compliance
- Managing approvals
This makes regulatory affairs careers highly valuable and in demand.
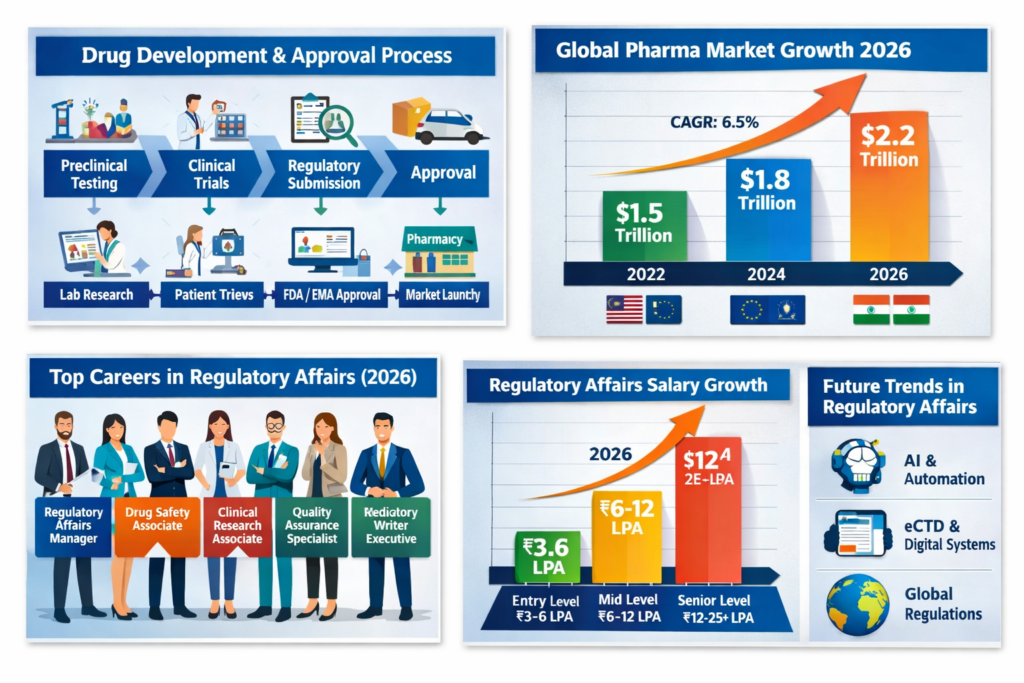
 Future Trends in Drug Approval
Future Trends in Drug Approval
The drug approval process is evolving with:
 AI in clinical trials
AI in clinical trials Digital submissions (eCTD)
Digital submissions (eCTD) Global harmonization of regulations
Global harmonization of regulations
These advancements are making the process faster and more efficient.
 Conclusion
Conclusion
The step-by-step drug approval process is a critical pathway that ensures every medicine reaching patients is safe and effective. From discovery to market launch, each stage involves strict evaluation and compliance.
For students and professionals, understanding this process opens doors to exciting careers in drug regulatory affairs, clinical research, and pharmacovigilance.
If you aim to build a future in the pharmaceutical industry, mastering this process is your first step toward success 






