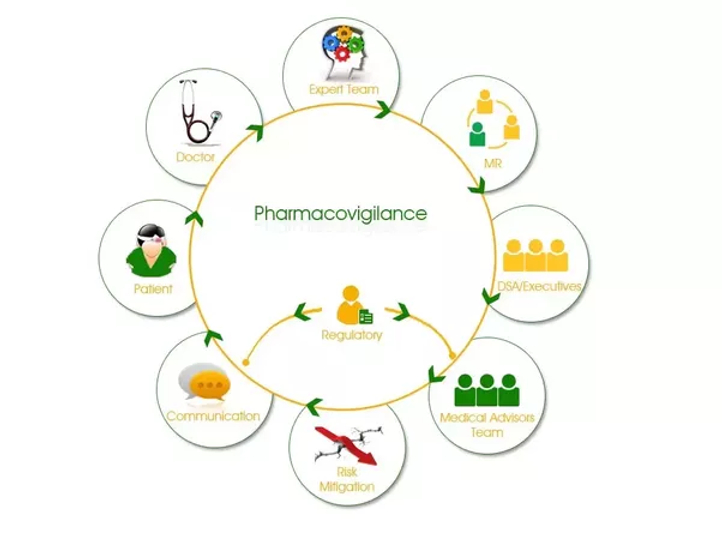
Change Management
Hello Readers, Hope it was a great weekend. And now as the night grows darker, and the day prepares to change from Sunday to Monday, there’s this little anxiety of the change. Talking of the change, let us now dig a deeper in the change management requirements during project management or in any industry […]