Computer Software Assurance (CSA)V/S Computer System Validation — And Why CSV Still Matters More
As digital systems continue to evolve, regulated industries such as pharmaceuticals, medical devices, and life sciences are rethinking how they ensure software quality and compliance. Traditionally, Computer System Validation (CSV) has been the backbone of compliance. More recently, regulators like the FDA have introduced odern, risk-based alternative. Computer System Validation (CSV) Computer System Assurance Computer […]
Data Migration Using CSV: A Structured and Risk-Based Approach

Introduction In today’s digital world, data migration is a regular part of system upgrades. CSV files are often the first choice because they are simple, lightweight, and work with almost every tool. But moving data using CSV still needs proper planning and validation, otherwise small mistakes can turn into big problems later. HOW DATA MIGRATION […]
The Role of LIMS in Ensuring Regulatory Compliance
Introduction In today’s highly regulated laboratory environment, maintaining regulatory compliance is essential for ensuring data accuracy, operational integrity, and public trust. Laboratories operating in industries such as pharmaceuticals, biotechnology, clinical diagnostics, food safety, and environmental testing must comply with stringent regulations and standards. A Laboratory Information Management System (LIMS) plays a critical role in helping […]
Why ER/ES Is Required in Computer System Validation (CSV)
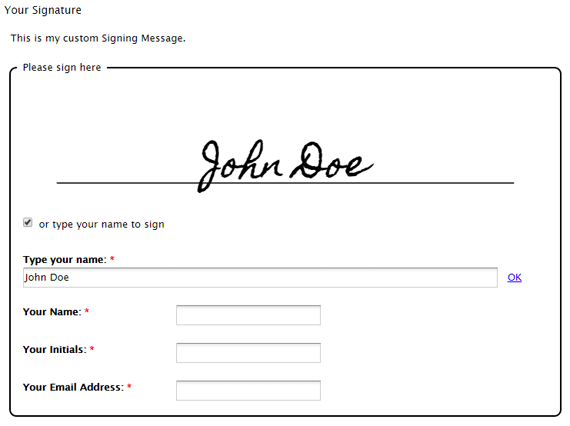
Computer System Validation (CSV) plays a critical role in regulated industries such as pharmaceuticals, biotechnology, and medical devices. Ensuring that computer systems perform as intended and comply with regulatory requirements is essential for product quality and patient safety. Within this process, Electronic Records (ER) and Electronic Signatures (ES) have become indispensable components. This post explores […]
Why CSV Matters in Regulated Industries

Introduction Computer System Validation consists of the activity necessary to confirm that computer systems perform their intended functions reliably, accurately, and consistently in a regulated environment. The testing and documentation of software and hardware systems are included in this, ensuring they meet predefined specifications and regulatory requirements. CSV is not only meant to confirm system […]
Change Control in CSV: Handling System Updates Without Risk

Introduction Change control is one of the most important elements of CSV, which provides assurance that changes to validated systems are examined systematically, documented, and implemented without compromising the system’s integrity, compliance status, or intended function. In regulated industries like pharmaceuticals, biotechnology, and medical devices, where computerized systems manage data impacting product quality and patient […]
Validating Excelsheets: Effective Approaches to Improve Data Accuracy

Data integrity is everything in today’s modern world. Whether it deals with financial reports, sales forecasts, or inventory tracking, several business outcomes are hinged on data accuracy. Excel sheets, being versatile, are used across industries to handle data. However, human error and incorrect formulas and inconsistency in data entry can jeopardize the accuracy of the […]
Continuous Validation: Is It the Future of CSV?
Introduction Explanation of Continuous Validation (CV) and its relevance in the context of Computer System Validation (CSV). Continuous Validation (CV) is an advanced approach to ensuring that computerized systems remain in a validated state throughout their lifecycle. Unlike traditional Computerized System Validation (CSV), which follows a one-time validation process at a specific point in time, […]
Transforming Business for Good: The Intersection of Digital Innovation and CSV
Introduction Digital transformation represents a strategic shift in how organizations operate, compete, and deliver value by leveraging advanced technologies. It’s more than the adoption of new tools—it requires a fundamental rethinking of traditional processes, customer engagement, and decision-making. With technologies such as cloud computing, artificial intelligence (AI), automation, and data analytics, businesses are streamlining workflows, […]
What Is FDA 21 CFR Part 11? A Guide for Computer System Validation Professionals

Introduction FDA 21 CFR Part 11 is a key regulation that governs the use of electronic signatures and electronic records in the pharmaceutical, biotech, medical device, and other FDA-regulated industries. Initially published in 1997, this regulation ensures that electronic records and electronic signatures are as trustworthy, dependable, and equivalent to handwritten signatures and paper records. […]
